6. Research and Innovation
You have a right to:
Be informed about ongoing research relevant to you, and your ability and eligibility to participate in research.
Three key questions that every cancer patient may choose to ask:
- Is our cancer service active in performing cancer research, enrolling patients in clinical trials and supporting innovation?
- Is there a study or clinical trial in which I could participate if I am eligible and if I choose to do so?
- May I go to another hospital or centre if I wish to be involved in a specific clinical research project not offered at my hospital or centre?
Explanation
European cancer patients should be informed about any ongoing research and innovation in the cancer service which is providing their care, while recognising that most patients will receive standard-of-care treatment. Research and innovation have underpinned improvements in outcomes for cancer patients in recent decades, with long-term survival increasing to over 50% of cancer patients in many European countries. Further improvements will depend substantially on appropriate implementation of research and innovation discoveries. If relevant clinical trials are available in another hospital which are not being offered in the patient’s hospital, patients should have the option to change hospital in order to participate. Patients should have the right to participate (or refuse to participate) in clinical research. The informed decision that they take, after careful discussion with the clinical investigators, is a personal one. Patients must also be assured by the clinical team that a decision to not participate in clinical research or to withdraw from a research study, will not adversely affect their care.
Clinical cancer research provides the evidence which ensures patients receive the best available care. It improves healthcare delivery, leading to better outcomes and improved quality-of-life for patients. Research which links the laboratory to the clinic is an essential component of a comprehensive cancer control strategy. There is evidence that research-active hospitals provide better care, achieve higher levels of patient satisfaction and deliver improved survival for patients compared to hospitals that are not active in research.
There are many kinds of clinical research. In some, researchers will ask for a patient’s consent just to collect information about them for research purposes. In other research, the patient’s diagnostic tests and treatments may be directly determined by clinical research protocols, but only after the patient has been fully informed and has given their consent. Patients may be asked to consider whether they are prepared to receive a new experimental treatment, in addition to or after they have received the standard care. They may also be invited to join in a randomised controlled trial (RCT). Here, one group of patients receive the standard-of-care treatment, while a second group of patients are allocated to a different treatment that is under trial. The process of allocation to the groups is called randomisation and it ensures that the two groups are truly comparable so that the results achieved are robust. At the end of the trial, results for the two groups of patients are compared so that the better treatment can be identified with confidence and used in future to improve cancer care.
Clinical research is highly regulated and carefully monitored to ensure that it is properly conducted, and that patient participation is properly sought with written informed consent and appropriate patient information. Very importantly, it is strictly monitored to ensure the maximum possible safety for patients who choose to participate in research studies.
Supporting Literature and Evidence
The Nature and importance of Translational Cancer Research
The progress made in characterising the basic biology of cancer and in developing new scientific knowledge and technology in physical sciences and engineering that are highly relevant to approaches to cancer diagnosis and treatment are very promising. However, to bring these discoveries into clinical practice requires a concerted and structured effort in what is known as translational cancer research and this can be challenging. First, basic research must be translated into a form that can be implemented in the clinic. This move from “bench to bedside” is often referred to as the “first translational gap”. Second, innovation that is shown to be valuable in clinical trials, must be taken up and disseminated across a whole healthcare system. This is often referred to as the “second translational gap”. Increasingly, the focus of cancer research and cancer research funding bodies in recent years has been on the potential for bringing new discoveries to the patient and a systematic and structured approach to timely translation into healthcare (18-20,144).
The impact of positive clinical trials on long term patient outcomes
Clinical trials are designed to determine if a new treatment is better than or equivalent to existing treatments or diagnostic strategies. When trials are positive and identify better care options, then this should lead directly to improve patient outcomes. It is important to note that even when new approaches to care are shown to be equivalent to existing approaches, this can still result in better care if the new approach is less toxic, less inconvenient or places less pressure and cost on the healthcare system. One of the most comprehensive evaluations of the benefits of positive clinical trials came from the US Southwest Oncology Group (SWOG) (145). Treatment trials during SWOG's 60-year history for which the new, experimental therapy provided a statistically significant improvement in overall survival were evaluated. It was assumed that the new, proven treatments from these trials established new standards for cancer care in the treatment community. They estimated population life-years gained from 23 positive treatment trials through 2015 by mapping the effect of the new treatments onto the US cancer population (Figure 18).
In total, 12,361 patients were enrolled to the 23 positive trials from 1965 to 2012. The study estimated that 3.34 million (95% confidence limit, 2.39 – 4.15 million) life-years were gained from these 23 trials through 2015 by the implementation of the results across the whole population. The US dollar return on investment was $125 per life-year gained. Although it is possible that in some cases the uptake of the new evidence may have been slower and less complete than had been modelled, these data still show the benefit for cancer patients, with an impressive return on investment in lives saved for the money spent (145).
Evidence from clinical trials may diffuse very slowly into daily clinical practice. There may be a number of reasons for this, such as insufficient knowledge diffusion, insufficient resources, and inadequate reimbursement systems. Examples include hypo fractionated RT for breast cancer (104), the use of single radiotherapy fractions for the palliative treatment of uncomplicated bone metastases (105-107).
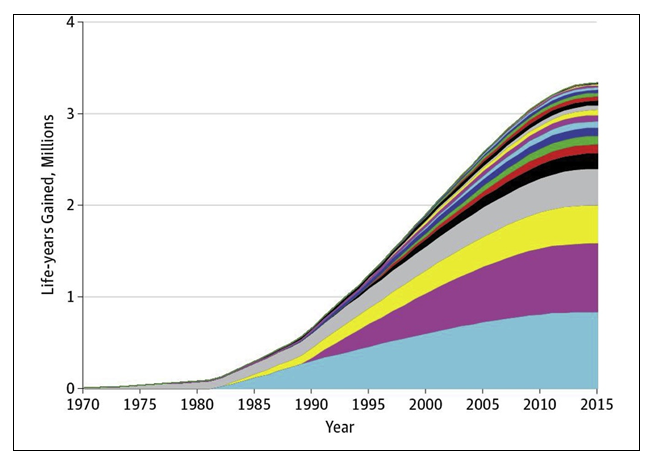
Figure 18. Cumulative life-years gained through 2015 by SWOG studies. The cumulative life years gained through the implementation of positive RCTs is plotted for each trial through to 2015. The colour coded areas represent cumulative life-years for each of the 23 studies evaluated. For each colour, the cumulative life years saved are plotted 1970–2015. The impact of each individual study is added with a new coloured segment so that the total cumulative, additive impact of all 23 studies combined is shown with the contribution of each study shown by a separate colour. Four studies are shown to contribute two thirds of the life years saved. Reproduced with permission (145)
The impact of research participation by healthcare institutions on patient outcomes
It has been claimed that patients who participate in clinical trials have better outcomes than patients in the same institutions who are not in trials. Some early studies appeared to support this view, but trial participants are a selected group of patients who tend to be younger and fitter than most cancer patients, so systematic reviews do not support this assertion concerning better outcomes (146).
A different and more pertinent question is to ask whether a clinical team or hospital which is research-active and participates actively by entering patients into trials in large numbers, delivers better outcomes for all the patients treated by that hospital or multidisciplinary team, when compared to the outcomes for a research-inactive hospital or team. Research participation might stimulate teams to consider new evidence, introduce new improved cancer treatments and equipment, and improve the quality assurance of treatments and investigations. If such improvements are stimulated by research participation, they should improve decision-making and have an impact on all treated patients with the disease, or even a wider group of patients with other conditions. The association between research activity and healthcare performance has recently been evaluated in systematic reviews which support the positive influence of research participation on the processes of care delivery (147). Clinical trial activity also correlates with improved care quality in hospitals judged by the UK Care Quality Commission (148-149).
A small number of studies have investigated whether a hospital's research activity in clinical trials is associated with improved survival for all of their patients with the disease studied (150). Studies in multiple myeloma, testicular cancer, coronary artery disease and epithelial ovarian cancer, all highlight positive associations between research participation and patient survival, but studies to date are too small in size and number to be conclusive. Modern information systems now allow us to examine the impact of research in greater detail and much larger numbers. In 2001, the National Institute for Health Research Cancer Research Network (NCRN) was established to provide the English NHS with the clinical infrastructure required to improve the recruitment, speed, quality and integration of clinical cancer research in all parts of the NHS, involving all hospitals in clinical cancer research. This approach converted much of the NHS to engage in research-intensive participation. Over the same period, the NHS in England collated and combined existing datasets to create comprehensive individual-patient records of cancer diagnoses, demographics, treatments and mortality. Linking cancer registry data, hospital episode data and clinical trials datasets became possible under rigorous conditions of confidentiality. The NCRN studies of colorectal cancer (151) showed, in a multivariable case mix adjusted analysis of over 200,000 patients, that there was a strong association between research participation and patient survival. A literature meta-analysis of studies which report the impact of research participation on the survival of patients within research-intensive hospitals, compared to those that were less research-active, supports the conclusion that the research-intensive hospitals have substantially better outcomes, independently of their size (150, 151).
Clinical trials and patient satisfaction with care
The UK National Cancer Patient Experience Survey (NCPES) of 60,000 patients, showed clear association between research participation and higher levels of patient satisfaction with care (152), Evidence also suggests that patients in more research-active hospitals have more confidence in staff and are better informed about their care (148).
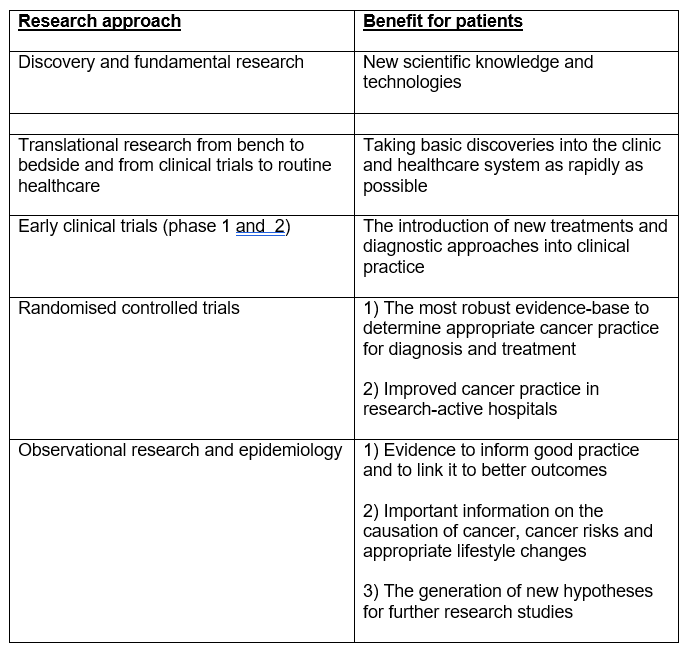
Table 9. Research benefits